CASE STUDY: centiTMF
The “Messy Middle” of
Compliance Intelligence.
Clinical trials run on paperwork, yet nobody knows if it's truly complete until it's too late. With centiTMF, we built a system to ensure inspection-readiness before auditors show up.
The Core Challenge
“In compliance, an unexplainable flag is worse than no flag at all. Initially, the LLM could do the heavy lifting but failed to explain its decisions, creating ‘hallucinated’ urgency that eroded user trust.”
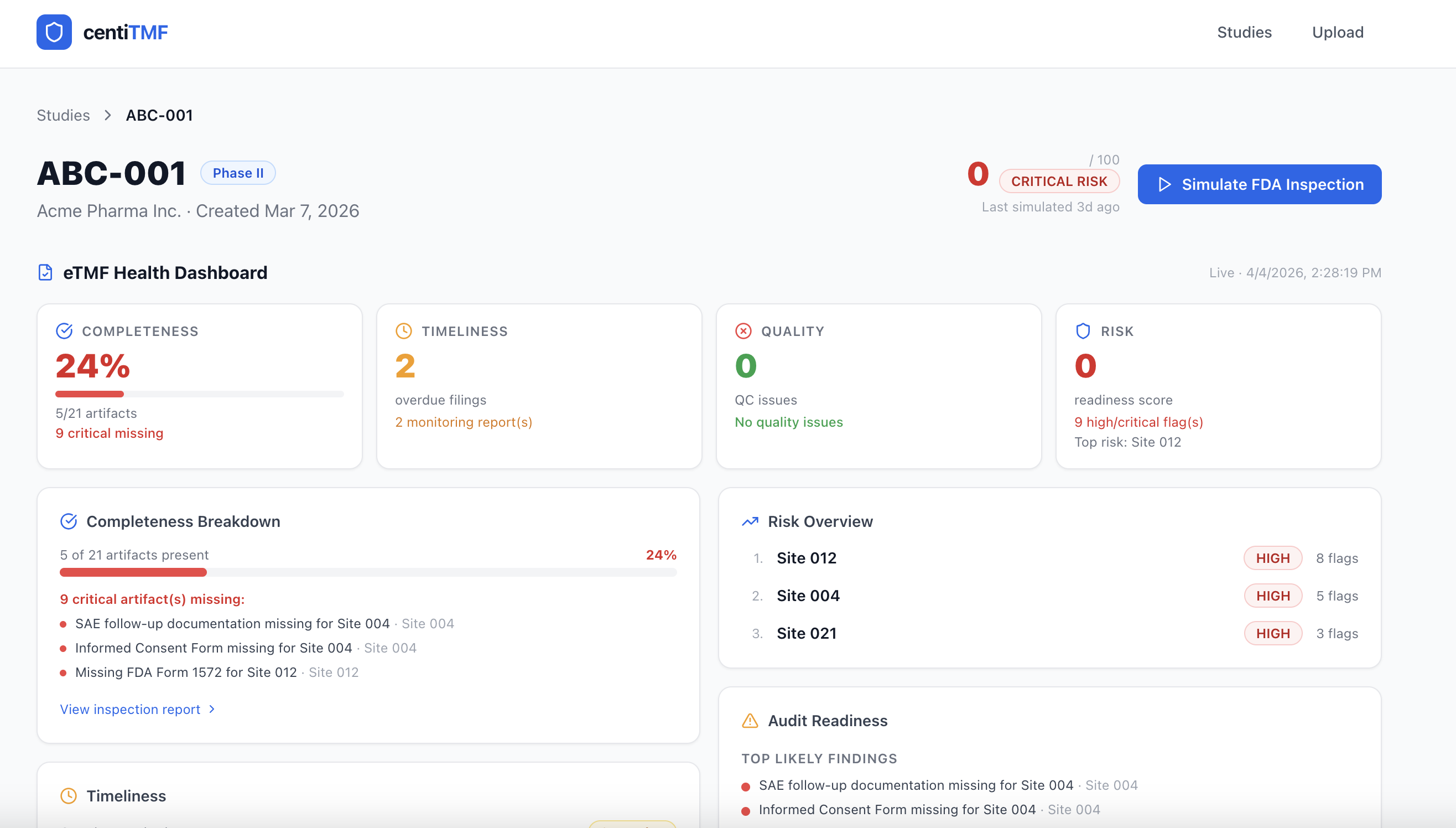
0–100
Readiness Score
100%
Traceability

All Case Studies
Ready for your next Deployment?
We are currently accepting high-priority consulting engagements.
Start a Conversation